KOLs Discussing lidERA
lidERA Key Slides & Visuals
Slides shared by KOLs at ASCO 2026 (Oral Abstract 502, Schmid). Click any image to expand.
Top Tweets on lidERA

Day 2 #ASCO26 highlights: Breast Cancer: 1. #KN522 (update): IO + Chemo TNBC 2. #OPTIMA: Adj Chemo HR+ 3. #lidERA: Adj SERD HR+ 4. #SENOMAC: ALND omission Prostate Ca 5. #TALAPRO3: PARPi NSCLC: 6. #TRITON: Dual ICI mNSCLC #OncTwitter @ASCO @OncoAlert 1/7 https://t

#ASCO26 Oral SERDs moving earlier in breast cancer? The phase III lidERA trial shows adjuvant giredestrant improved outcomes vs standard endocrine therapy across BOTH premenopausal and postmenopausal HR+/HER2− early breast cancer. 🔹 Premenopausal: • IDFS HR 0.65 • 42% https://

Benefit of adjuvant giredestrant over SoC endocrine treatment was observed irrespective of menopausal status, with a trend towards larger benefit in premenopausal patients. Comparable tox with giredestrant vs AI also seen irrespective of menopausal status (ie unrelated to OFS) ht

💫🌟🚨 Top 10 #BreastCancer abstracts for #ASCO26 — selected by our leads and finalized via a Delphi voting process 🗳️🔬 1️⃣ 500 — OPTIMA 2️⃣ LBA1006 — PERSEVERA BC 3️⃣ 507 — KEYNOTE-522 final analysis 4️⃣ LBA1007 — SERENA-6 5️⃣ 502 — LIDERA BC 6️⃣ LBA1000 — ASCENT-04 7️⃣ 501 — NATA

#asco26. Key data from the exciting LidERA study in premenop pts presented by Schmid. Benefit x menopausal status & most Rx with tam in control received LHRHa. Less d/c due to pain. Longer FU needed for medium risk. Q - combo with CDK4/6i or instead? More data T/F @OncoAlert

#ASCO26 In lidERA, adjuvant giredestrant maintained its IDFS/DRFI benefit regardless of menopausal status. Oral SERDs are entering the adjuvant HR+/HER2− space. However, the optimal strategy remains unclear: upfront oral SERD, a switch strategy after 2–3 years of AI, or https:

ASCO 2026 Update — lidERA Trial Menopausal Subgroup Analysis • Giredestrant improved IDFS and DRFI across both menopausal groups • Strongest signal seen in PRE-menopausal patients • ~42% relative reduction in recurrence risk in PRE-menopausal subgroup • Particularly notable http

lidERA BC #ASCO26 Giredestrant (oral SERD) demonstrated consistent IDFS benefit over standard-of-care endocrine therapy in ER+/HER2− early breast cancer — regardless of menopausal status. IDFS HR: Pre-menopausal: 0.65 (3-yr rate: 94.0% vs 91.5%) Post-menopausal: 0.74 (3-yr ht

Beautiful discussion by Lisa Carey, putting lidERA into context @OncoAlert #SABCS25 https://t.co/cY8MflK9a3

#SABCS25 Recap #2 | #lidERA The lidERA trial showed adjuvant oral SERD #giredestrant was superior to SoC ET (AI or TAM) in intermediate–high risk HR+ HER2– EBC. Here is my take: 1️⃣ A true milestone After ~20 years, we finally have a better-tolerated adjuvant ET: https://t.co

Oooh boy lidERA data looks good! I’m excited!!! A more tolerable endocrine therapy and more effective!!! #SABCS2025 https://t.co/pewJHHb2Fz

Giredestrant reduces the risk of invasive disease recurrence or death by 30% (6.7% vs. 9.4% events) compared with SOC ET in early breast cancer @OncoAlert @SABCSSanAntonio @dradityabardia #SABCS25 https://t.co/ar59Md8pGd
Overview
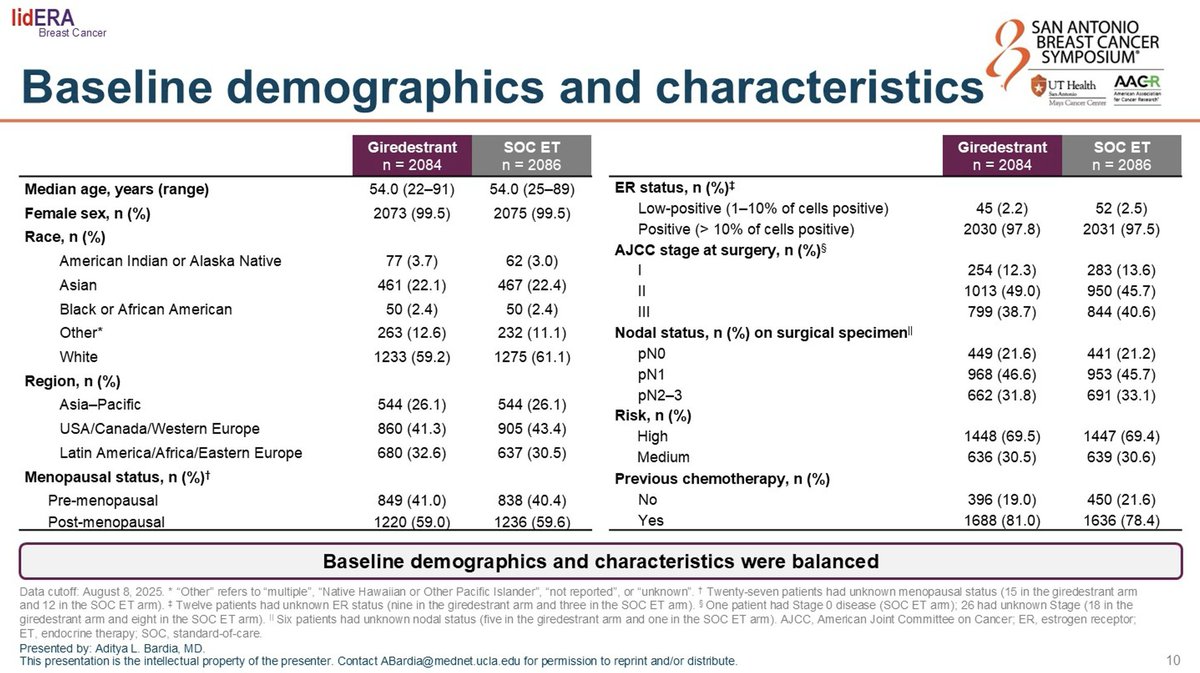
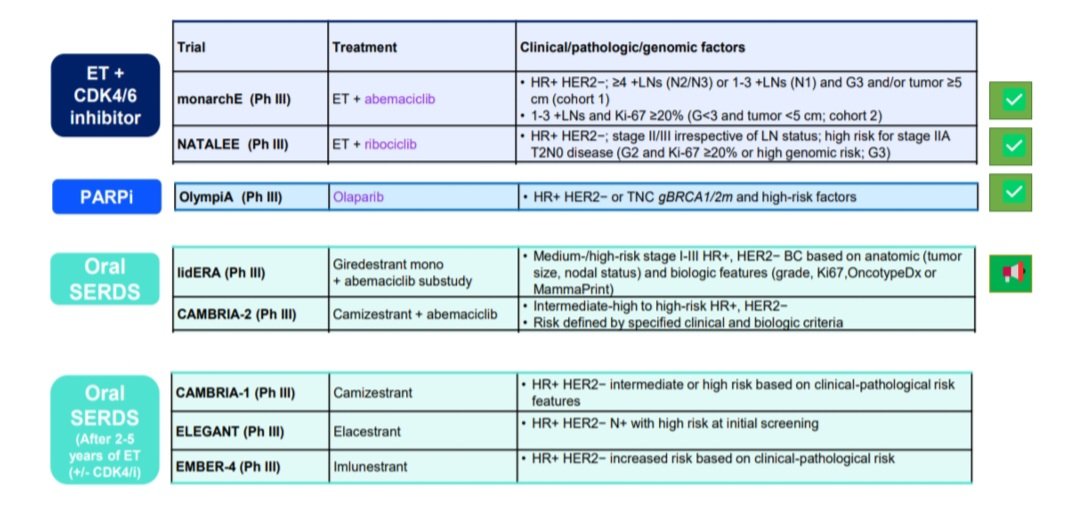
lidERA BC is a Phase 3 global randomized trial evaluating adjuvant giredestrant, an oral selective estrogen receptor degrader (SERD) from Roche/Genentech, versus investigator's choice of standard-of-care endocrine therapy (aromatase inhibitor or tamoxifen, plus an LHRH agonist in pre-menopausal women) in patients with stage II/III HR+/HER2− early breast cancer. The primary endpoint is invasive disease-free survival (iDFS); key secondary endpoints include distant recurrence-free interval (DRFI), overall survival, and safety.
Conference history: The lidERA primary readout (GS1-10) was first presented at SABCS 2025 (Dec 2025) by Aditya Bardia, MD MPH (UCLA Jonsson Comprehensive Cancer Center). The ASCO 2026 update (Abstract 502, May 30, 2026) presented by Peter Schmid reported the menopausal subgroup analysis, confirming that the iDFS benefit holds across pre- and post-menopausal patients.
Study Design
Phase 3, global, randomized, open-label trial comparing adjuvant giredestrant vs investigator's choice endocrine therapy. ~41% of patients enrolled were pre-menopausal.
Population
Stage II/III HR+/HER2− early breast cancer in patients who completed locoregional treatment. Both pre- and post-menopausal women were enrolled.
Intervention
Experimental: Adjuvant giredestrant (oral SERD). Control: Investigator's choice of AI or tamoxifen, plus LHRH agonist in pre-menopausal women.
Endpoints
Primary: Invasive disease-free survival (iDFS). Secondary: Distant recurrence-free interval (DRFI), overall survival, safety. ASCO 2026 reported menopausal subgroup analysis.
Reported Results (ASCO 2026 Subgroup Analysis)
Overall iDFS — SABCS 2025 Primary Endpoint (Bardia GS1-10)
3-year iDFS 92.4% with adjuvant giredestrant vs 89.6% with SoC endocrine therapy (HR 0.70; 95% CI 0.57–0.87; p=0.0014) — a 30% reduction in invasive disease recurrence or death at the pre-specified interim analysis. n=4,170 randomized 1:1; data cut-off Aug 8, 2025; median follow-up 32.3 months. Stage subgroup HRs: stage II HR 0.58 · stage III HR 0.74.
3-yr iDFS 92.4% vs 89.6% · HR 0.70 (CI 0.57–0.87) · p=0.0014Source 1: Roche SABCS 2025 press release (Dec 10, 2025)Source 2: The ASCO Post recap (Bardia GS1-10)
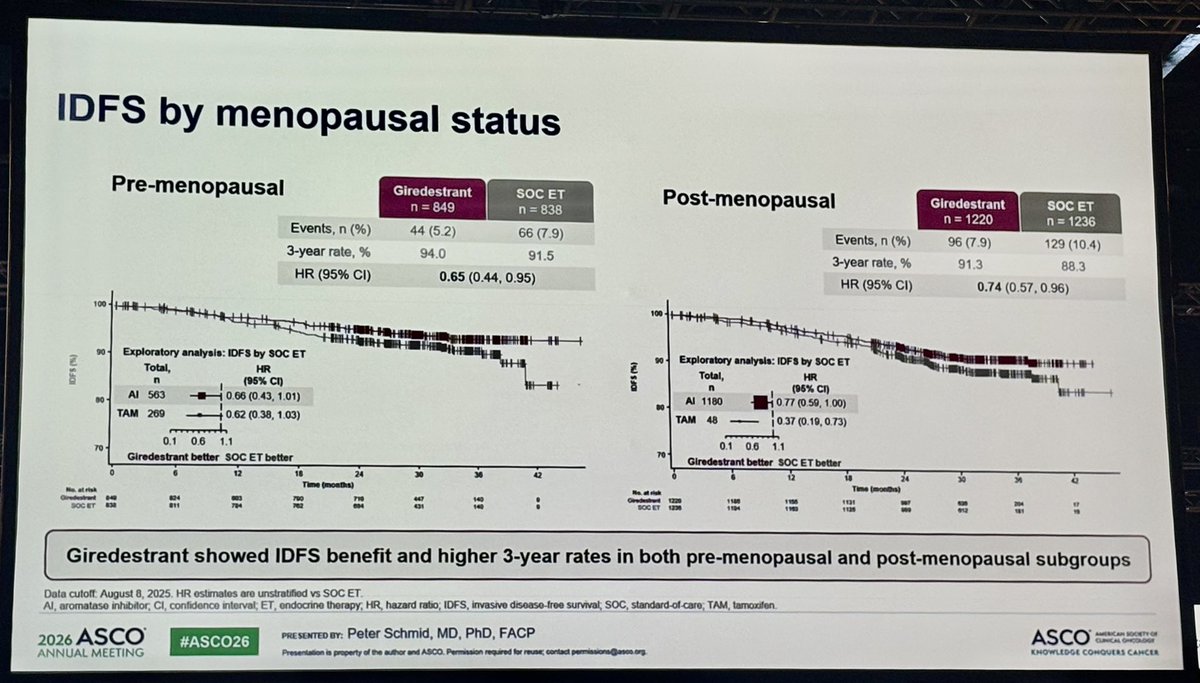
3-Year iDFS — Pre-menopausal Subgroup (ASCO 2026 Abs 502)
3-year iDFS was 94.0% with adjuvant giredestrant vs 91.5% with SoC endocrine therapy (HR 0.65), corresponding to a 42% relative reduction in invasive disease risk in pre-menopausal patients.
Pre-menopausal 3-yr iDFS 94.0% vs 91.5% · HR 0.65 · 42% reductionSource 1: DocWire News — ASCO 2026 Abs 502 (Schmid menopausal subgroup)Source 2: Roche ASCO 2026 prep release (May 19, 2026)
3-Year iDFS — Post-menopausal Subgroup (ASCO 2026 Abs 502)
3-year iDFS was 91.3% with adjuvant giredestrant vs 88.3% with SoC endocrine therapy (HR 0.74), a 24% relative reduction. Consistent benefit across menopausal status with a numerically larger delta in the pre-menopausal cohort.
Post-menopausal 3-yr iDFS 91.3% vs 88.3% · HR 0.74 · 24% reductionSource 1: DocWire News — ASCO 2026 Abs 502Source 2: Roche ASCO 2026 prep release
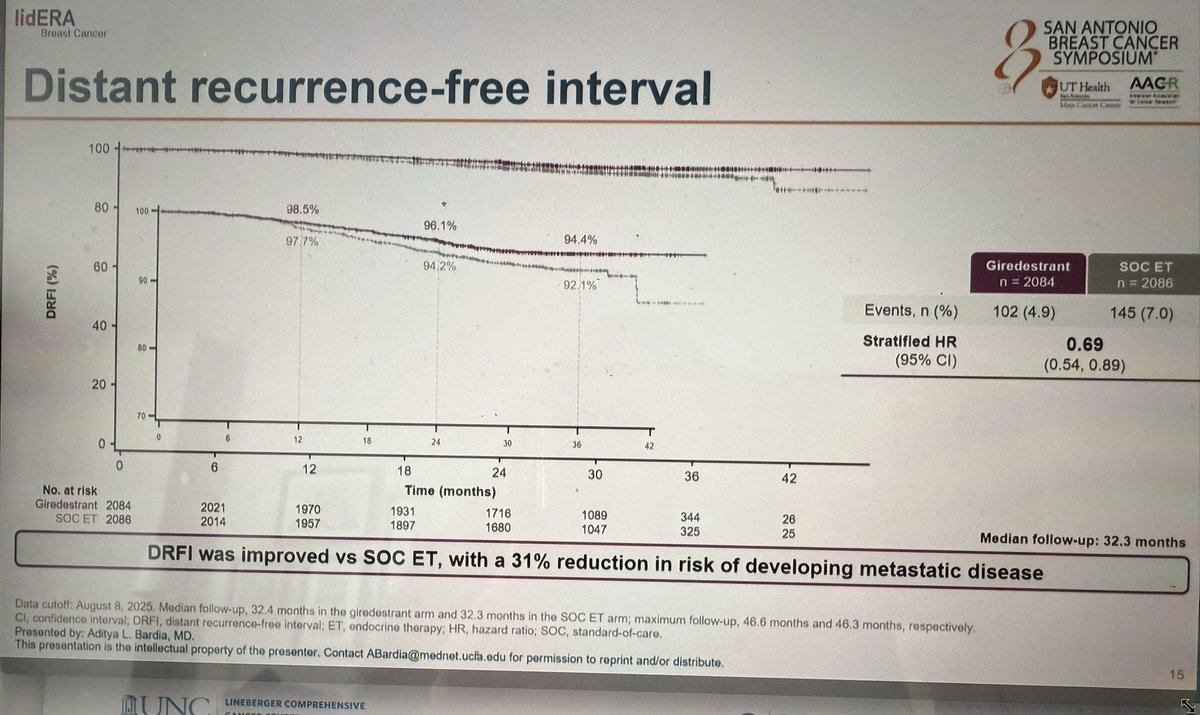
Distant Recurrence-Free Interval (DRFI)
Overall DRFI HR 0.69 (95% CI 0.54–0.89); 3-yr DRFI 96.1% vs 94.2% (SABCS 2025 primary). By menopausal status (ASCO 2026): Pre-menopausal DRFI 95.5% vs 92.6% (HR 0.58); Post-menopausal DRFI 93.4% vs 91.4% (HR 0.76).
Overall DRFI HR 0.69 · Pre HR 0.58 · Post HR 0.76Source 1: The ASCO Post (Bardia SABCS 2025 — overall DRFI)Source 2: DocWire News (ASCO 2026 Abs 502 — menopausal DRFI)
Safety & Tolerability
Treatment discontinuation rates: ~5.3% with giredestrant vs ~8.2% with SoC endocrine therapy — fewer all-cause discontinuations in the giredestrant arm, largely driven by lower rates of arthralgia / musculoskeletal AEs. Bradycardia 11.3% (giredestrant) vs 3.2% (SoC) — grade 1, asymptomatic, did not require interruption or discontinuation per Bardia; consistent with the known oral-SERD class effect. Per @ErikaHamilton9: "Giredestrant discontinuations were less than AI — largely due to fewer arthralgias (finally!!!)."
D/c 5.3% vs 8.2% · Bradycardia 11.3% vs 3.2% (G1, asymptomatic)Source 1: AACR / SABCS 2025 news release (Bardia)Source 2: SABCS Meeting News (General Session 1 official summary)
Open Clinical Questions
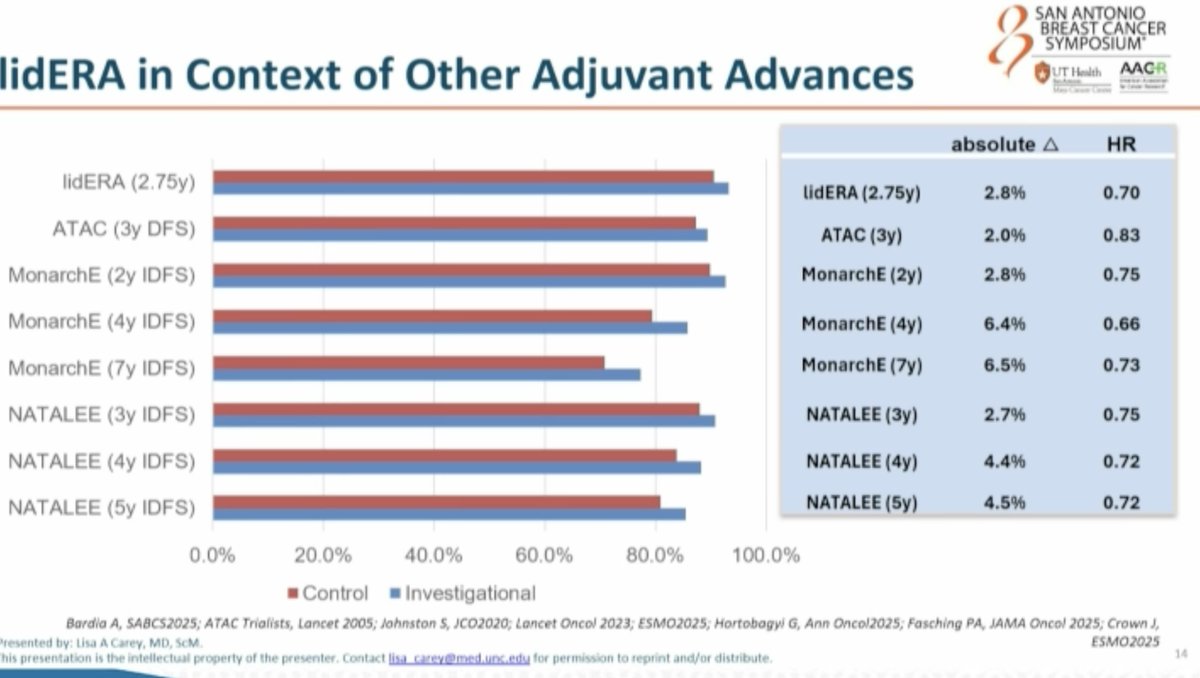
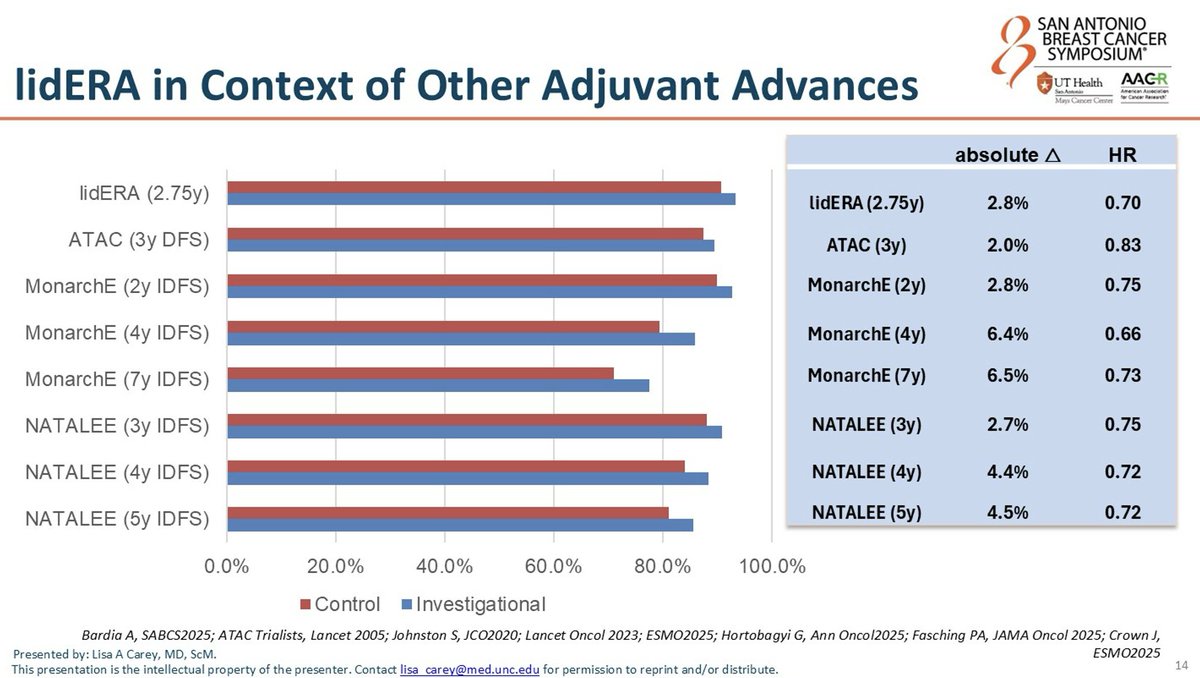
Per @dr_yakupergun and @ErikaHamilton9: oral SERDs are entering the adjuvant HR+/HER2− space, but the optimal strategy remains unclear — upfront oral SERD vs switch strategy vs combination with CDK4/6 inhibitors. lidERA pre-dated CDK4/6 adjuvant approvals and compared giredestrant vs investigator's choice ET (tamoxifen / letrozole / anastrozole / exemestane), NOT vs CDK4/6 + AI. Cross-trial HR comparison: lidERA HR 0.70 vs monarchE HR 0.65 (abemaciclib+ET) vs NATALEE HR 0.75 (ribociclib+ET). @hoperugo flagged that longer follow-up is needed for medium-risk patients.
Source 1: The ASCO Post (SABCS 2025 expert commentary)Source 2: ApexOnco cross-trial comparison (lidERA / monarchE / NATALEE)