GU Cancer Researchers Discuss Latest Breakthroughs at #GU25
Breakthroughs in Genitourinary Oncology: Insights from GU25 The GU25 conference delivered groundbreaking insights into the evolving landscape of...
4 min read
Brian Shields
:
Mar 29, 2025 6:40:06 AM

This is a collection of resources from the GU Oncology KOL Community with commentary and direct sources to key clinical trial presentation of the NIAGARA Trial Leading to FDA Approval for durvalumab for resectable MIBC.
JUST IN (again): @US_FDA approved Durvalumab for the Neoadjuvant treatment of muscle-invasive bladder cancer based on the #Niagara study https://t.co/GRXKnr4HuA
— Toni Choueiri, MD (@DrChoueiri) March 28, 2025
Study presented #ESMO24 /published in @NEJM https://t.co/J1Q7tbaFWt
HR EFS 0.68 (0.5-0.8)
HR OS 0.75 (0.59-0.93)
Durvalumab now @US_FDA approved in resectable MIBC based off #NIAGARA: Ph III, Gem/Cis +/- Durvalumab —> ✂️ —> post-op Durva
— Oncology Brothers (@OncBrothers) March 28, 2025
- New SoC
- 2 yr OS: 82.2% vs. 75.2% (HR: 0.75)
- 2yr EFS: 67.8% vs. 59.8% (HR: 0.68)
- Do we need adj Durva if pCR? #OncTwitter #MedTwitter #gusm pic.twitter.com/4Uia7MP3YE
NIAGRA presented #ESMO24 and published today @NEJM – huge accomplishment 1000+ patients enrolled. Exciting results!
— Elizabeth Plimack MD (@ERPlimackMD) September 15, 2024
Some thoughts on applying them to practice... pic.twitter.com/6vXd50lC83
Brilliant discussion by @PGrivasMDPhD about NIAGARA trials focused on different roles of neoadj vs. adjuvant part of the study and future perspectives opened by this study. @BladderCancerUS @OncoAlert @GUOncologyNow pic.twitter.com/xOfYTnGRYf
— Roberto Iacovelli (@DrIacovelli) September 15, 2024
⚡️UC Oral Abstract Session⚡️
— Zach Klaassen (@zklaassen_md) February 14, 2025
Additional outcomes and an exploratory analysis of the impact of pCR on long-term outcomes from NIAGARA @MattGalsky @urotoday #GU25
Durva vs Comparison Arm:
📌⬆️EFS (HR 0.68, 95% CI 0.56-0.82) ⬆️ OS (HR 0.75, 95% CI 0.59-0.93), 10% ⬆️ in pCR rate… pic.twitter.com/7GMS53H1Us
💫🌟🚨🔝🔝🔝💥 NIAGARA Trial Phase 3 Results! 💥@OncoAlert @myESMO
— Dra. María Natalia Gandur Quiroga (@nataliagandur) September 15, 2024
Presented by @tompowles1, at #ESMO24 on Durvalumab + Chemotherapy in Muscle-invasive Bladder Cancer (MIBC).
Key results:
🔹 Event-Free Survival (EFS) at 24 months:
Durvalumab Arm: 67.8%
Comparator Arm: 59.8%… pic.twitter.com/ML97Y03Ale
Valid points to show pitfalls in Niagara trial . The question of benifit of neoadjuvant IO over adjuvant IO alone is unanswered. As in tnbc or nsclc. @ASCO #GU25 pic.twitter.com/PY4OqV26Oh
— Dr Amol Akhade (@SuyogCancer) February 15, 2025
Where are post-recurrence data? Not available.
— Timothée Olivier, MD (@Timothee_MD) September 15, 2024
NIAGARA enrolled in LMIC countries with limited access to best post-recurrence care, which likely led to positive OS. Despite high expectations, NIAGARA did not have a standing ovation, and this is a good sign for oncology! https://t.co/8fXUNKohQ2 pic.twitter.com/ISWhYi4MWw

Breakthroughs in Genitourinary Oncology: Insights from GU25 The GU25 conference delivered groundbreaking insights into the evolving landscape of...
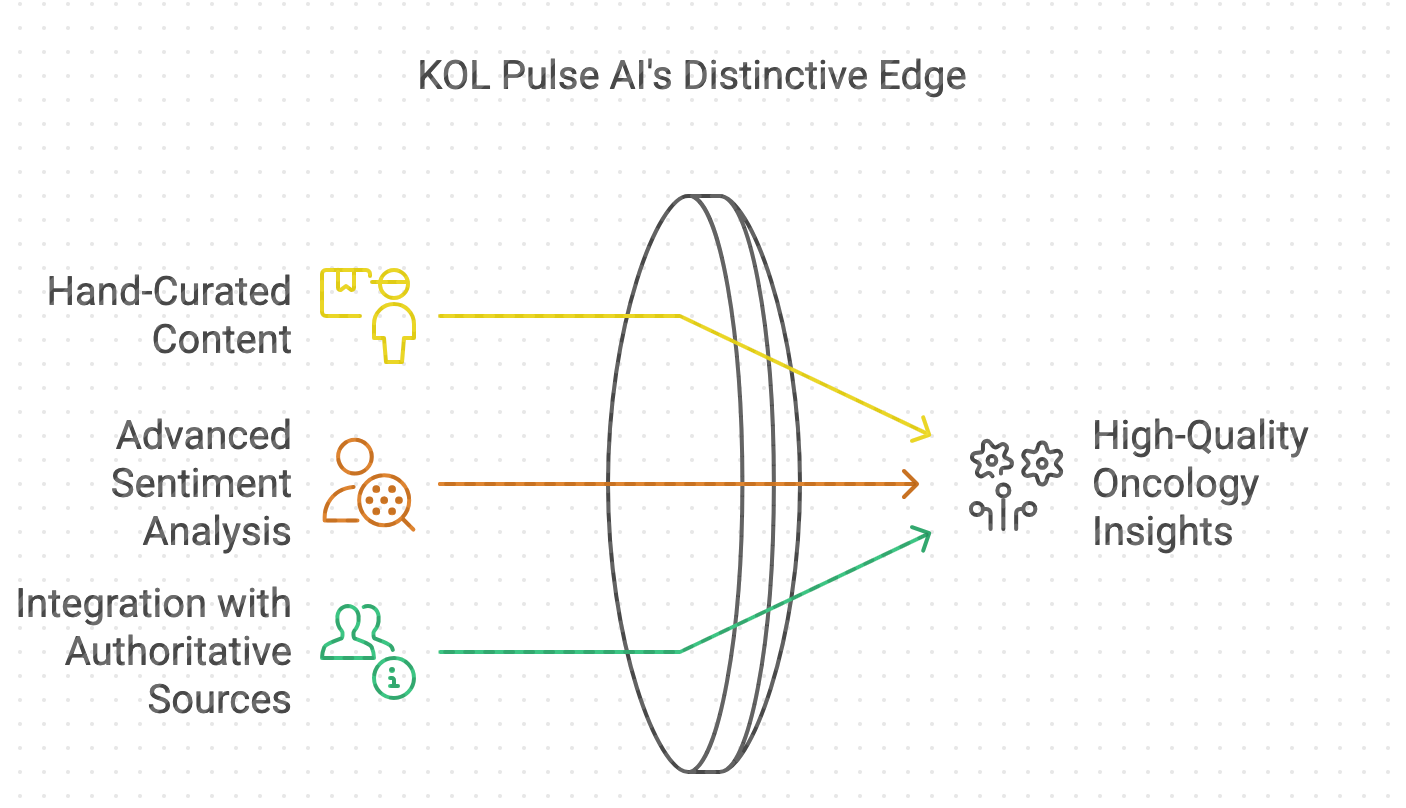
KOL Pulse AI is a specialized platform that leverages hand-curated Key Opinion Leader (KOL) conversations in oncology, enhanced by advanced AI...